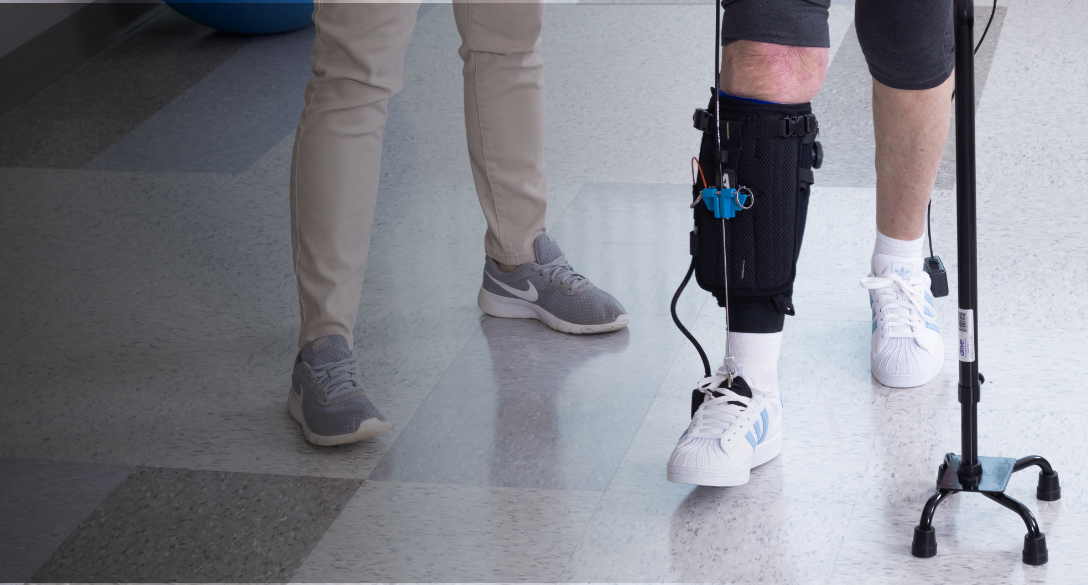
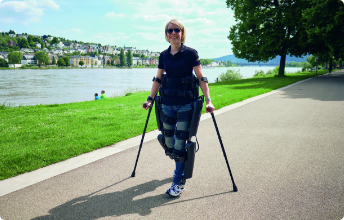
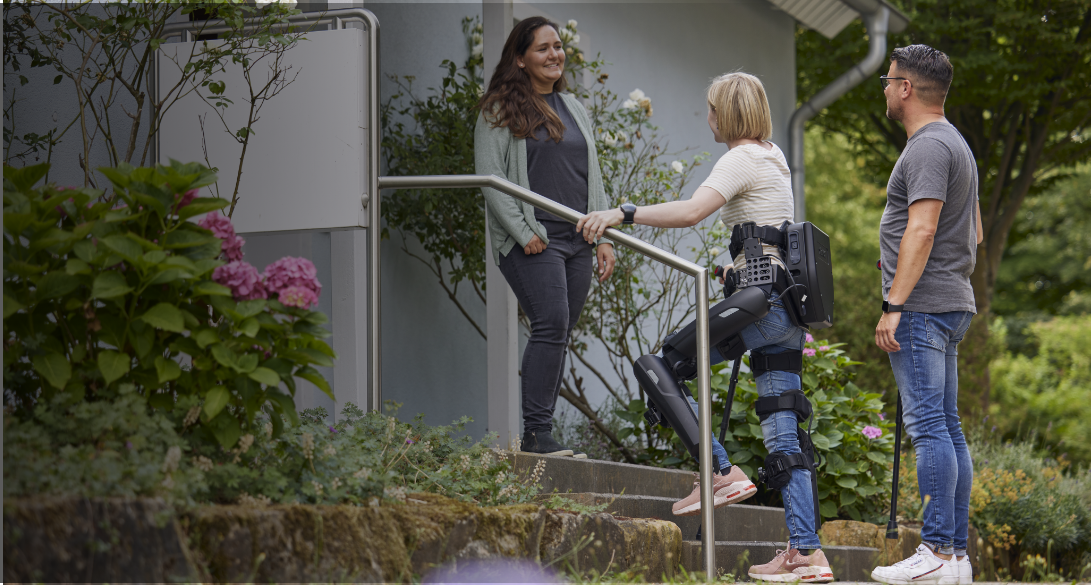Introducing Lifeward, previously ReWalk Robotics.
Our mission at Lifeward is to relentlessly drive innovation to change the lives of individuals with physical limitations or disabilities. We are committed to delivering groundbreaking solutions that empower individuals to do what they love.
As a global market leader in pioneering life-changing solutions, our portfolio features innovative products including the ReWalk Exoskeleton, the ReStore Exo-Suit, the MYOLYN FES bikes, and our newest addition, the AlterG Anti-Gravity systems. We will continue to be the driving force to elevate the standard of care in overcoming physical limitations and disabilities.
It’s not just upward. It’s Lifeward.